Pipeline

Nobelpharma committed to development of critical but neglected drugs in order to fulfill social mandate
We have constantly been aspiring to conduct research and development of drugs, that are not the prime targets for many pharmaceutical companies due to questionable economic viability with a small number of patients (drugs for unmet medical needs). Our development pipeline mainly includes the drugs such as orphan drugs (drugs for rare diseases) that have been strongly requested by patients, academic societies, or the government, as well as off-label drugs and pediatric medicines. The efficient research and development processes by our small but highly capable organization is what enables us to obtain approvals for such drugs with a comparatively small market size.
While we heretofore have engaged in the development of many unapproved drugs, that were already available in US and Europe but were not developed in Japan despite their significant need, we will also actively take on the challenges of research and development under new and different concepts.
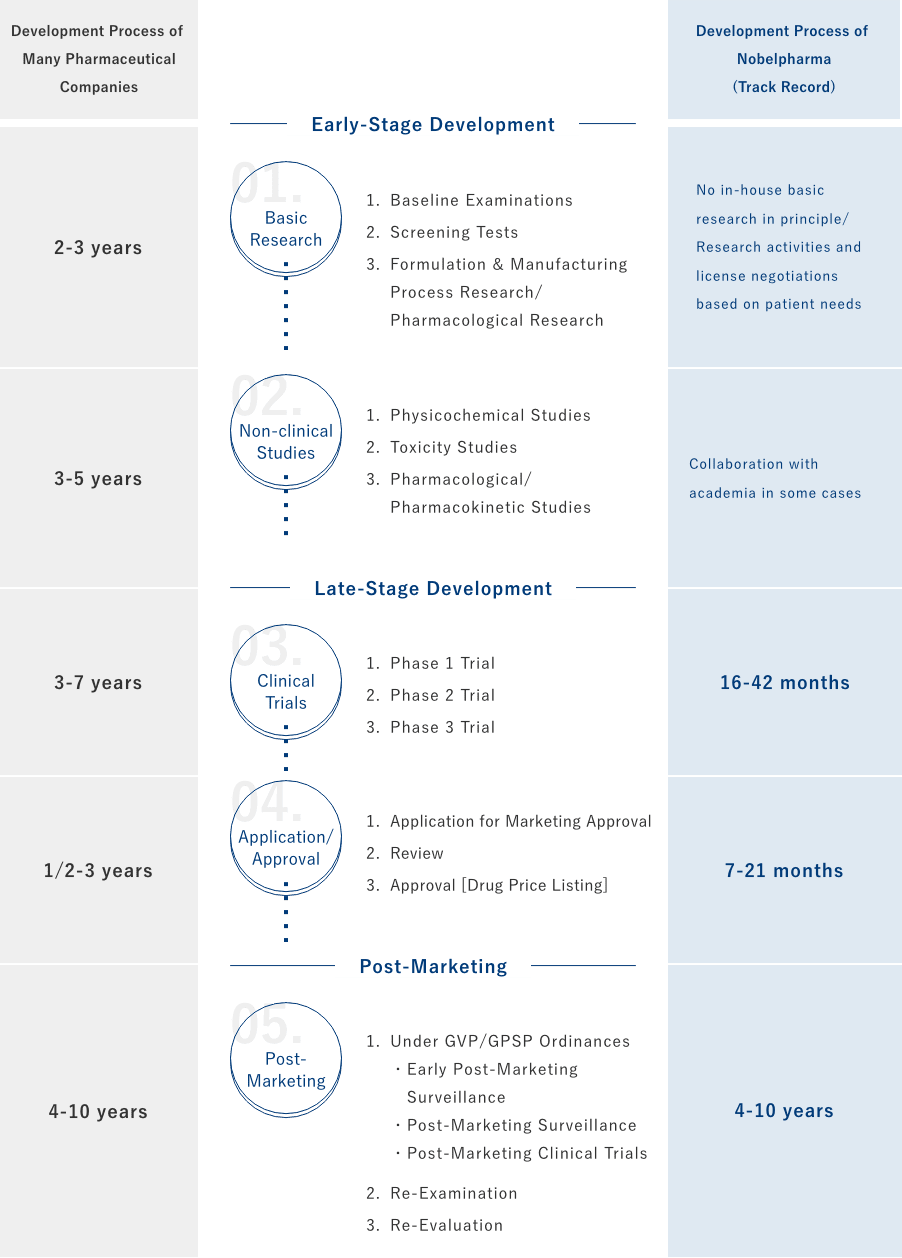
Development Process of Nobelpharma
Future Development Projects
The pipeline shows the developmental status of an unapproved drug or an unapproved indication and is not intended to promote or advertise any of the drugs.
- Note: Indicagtions and Approval are merely the expectations.
A.New Drugs/New Devices/regenerative medicine products (including global development projects)
As of January 2026
| Compound | Expected Indication |
Partner | Phase | Expected MA | |
|---|---|---|---|---|---|
| 1 | NPC-22 scopolamine |
hypersalivation | in-house | PII/III | Sep 2027 |
| 2 | NPC-33 naxitamab |
neuroblastoma | Y-mAbs Therapeutics |
PI | Dec 2027 |
| 3 | NPC-30 (GAIA-102) high-active NK-like cells |
neuroblastoma | GAIA BioMedicine Kyushu Univ |
PI | Feb 2028 |
| 4 | NPC-29 ubiquinol |
multiple system atrophy | Tokyo Univ | PIII in prep | Aug 2029 |
| 5 | NPC-32 platelet aggregation |
cardiovascular surgery/emergency care | National Defense Medical College Waseda Univ |
preclinical | - |
| 6 | MD-02 | Laser systems for ENT | A.R.C. Laser Gmbh. | filing | - |
B.Life Cycle Management (including global development projects)
As of January 2026
| Compound | Expected Indication |
Partner | Phase | Expected MA | |
|---|---|---|---|---|---|
| 1 | NPC-18 Retympa |
ear canal regeneration | Kaken | filing | Dec 2026 |
| 2 | NPC-12 Rapalimus |
primary immunodeficiency syndrome | Institute of Science Tokyo National Defense Medical College |
filing | May 2027 |
| 3 | NPC-25 Zintus |
hypozincemia (new formulation/additional dosage for pediatric) |
in-house | PIII | Sep 2027 |
| 4 | NPC-12 Rapalimus |
pure red cell aplasia | Shinshu Univ | PIII | Jan 2028 |
| 5 | NPC-06 Fostin |
trifacial neuralgia | Pfizer | PIII | Nov 2028 |
| 6 | NPC-15 Melatobel |
sleep-onset difficulty associated with mild cognitive disorder or dementia | in-house | PII | - |
| 7 | NPC-12 Rapalimus |
Pendred syndrome | Keio Univ Kitasato Univ |
PIII in prep | - |
| 8 | NPC-12 Raparimus |
generalized scleroderma | Oita Univ | PI/II | - |
| 9 | NPC-12G Rapalimus Gel |
vascular abnormality-associated skin lesions | Wakayama Medical Univ | PII/III in prep Multi-Regional Clinical Trial |
- |
| 10 | NPC-26 Sargmalin |
non-tuberculous mycobacterial disease | Niigata Univ Partner Therapeutics |
PII | - |
C.Overseas Development
As of January 2026
| Compound | Expected Indication |
Partner | Phase | Expected MA | ||
|---|---|---|---|---|---|---|
| 1 | NPC-02 (NOBELZIN) |
hypozincemia | in-house | CH | approved | Dec 2026 |
| 2 | NPC-31 (P092 maleic acid) |
prion disease | Gifu Univ | (Global) | PI/II in prep | - |
- Research and Development
